 Preeclampsia is one of the most serious complications of pregnancy. It usually appears after the 20th week and is characterized by high blood pressure, often accompanied by signs of damage in organs such as the kidney, liver, or brain. In severe cases, it can put both mother and baby at risk and may require premature delivery.
Preeclampsia is one of the most serious complications of pregnancy. It usually appears after the 20th week and is characterized by high blood pressure, often accompanied by signs of damage in organs such as the kidney, liver, or brain. In severe cases, it can put both mother and baby at risk and may require premature delivery.
Although symptoms appear later in pregnancy, the biological changes associated with preeclampsia may begin much earlier. Detecting these early signals could help improve risk assessment, monitoring, and clinical decision-making.
A molecular signal in maternal blood
In this study, published in Nature Communications, researchers from the Carlos Simon Foundation and iPremom analyzed cell-free RNA, or cfRNA, in maternal plasma. cfRNA consists of small RNA fragments released into the bloodstream by different tissues.
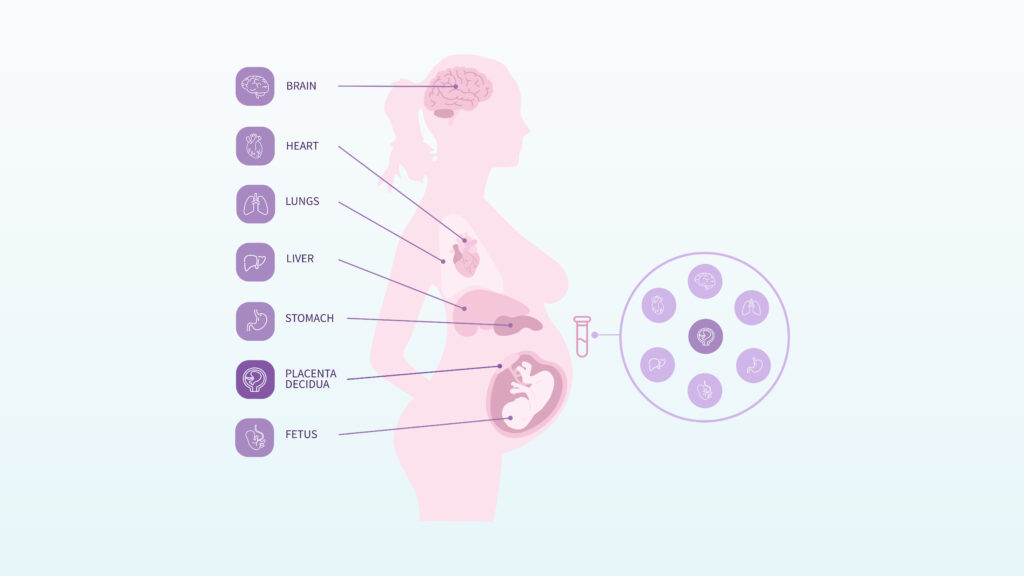
During pregnancy, these fragments can carry information from tissues involved in maternal and fetal health, including the decidua, placenta, liver, kidney, and immune system. In this way, a blood sample can offer a molecular snapshot of what is happening in the body before clinical symptoms become visible.
A large prospective pregnancy cohort
The case-control study was based on a prospective cohort of 9,586 pregnant women recruited across 14 hospitals in Spain. For the cfRNA analysis, the researchers selected 216 participants: 42 women with early-onset preeclampsia, 43 with late-onset preeclampsia, and 131 women with uncomplicated pregnancies.
Blood samples were collected at different stages of pregnancy, including the first trimester, the second trimester, and the time of diagnosis or later pregnancy. This allowed the team to compare molecular changes before and during the clinical onset of the disease.
Early and late preeclampsia showed different molecular patterns
The cfRNA models predicted early-onset preeclampsia from the first trimester, on average 18 weeks before clinical diagnosis. Prediction of late-onset preeclampsia was more limited in the first trimester, but improved in the second trimester.
The study also showed that early- and late-onset preeclampsia were not molecularly identical. Early-onset preeclampsia was strongly associated with signals from the maternal decidua, the uterine tissue involved in implantation and early placental development. Late-onset preeclampsia showed broader systemic signals from different tissues.
These findings support the idea that preeclampsia is not a single uniform condition, but a complex syndrome with different biological pathways.
What this could mean for pregnancy monitoring
Maternal plasma cfRNA can help identify pregnancies at higher risk before preeclampsia becomes clinically visible. This type of information supports closer monitoring, more personalized follow-up, and earlier preventive strategies when appropriate.
The findings also reinforce the importance of studying pregnancy complications from their earliest biological stages, before symptoms appear. Understanding these early molecular changes is a key step toward improving maternal and fetal health.
