Uterine leiomyomas, commonly known as fibroids, are the most frequent pelvic tumors in women of reproductive age. Up to 70% of women may develop them during their lifetime [1]. While many remain asymptomatic, approximately one in three patients experience symptoms such as heavy menstrual bleeding, pelvic pain, anemia, infertility, or recurrent pregnancy loss.
Fibroids are considered benign tumors. However, they can be difficult to distinguish from uterine leiomyosarcoma, a rare but aggressive malignant tumor arising from the same muscular layer of the uterus, the myometrium.
This overlap raises an important clinical question: can a fibroid become cancer?
Fibroids vs. Sarcoma: What Do We Know?
Current evidence suggests that uterine leiomyomas and leiomyosarcomas are biologically distinct tumors, with significantly different genomic and transcriptomic profiles [2].
In most cases where a sarcoma is diagnosed, it is believed to have been malignant from the outset, rather than having evolved from a pre-existing fibroid. However, definitive proof that malignant transformation never occurs is still lacking, so absolute statements should be avoided.
What is clear is the difference in prevalence: fibroids are extremely common, whereas leiomyosarcoma is rare [1,2].
The Intermediate Category: STUMP
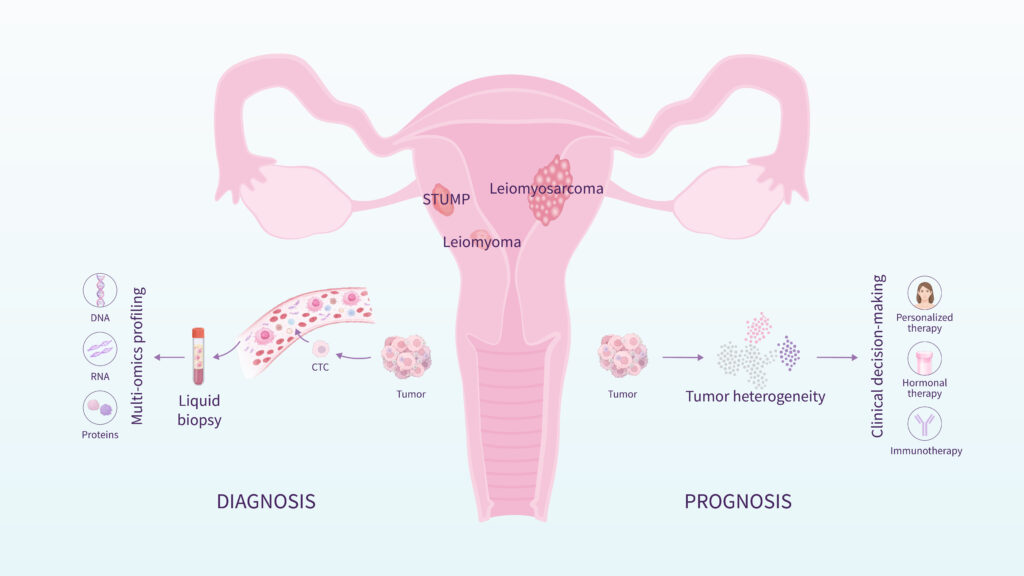
Between clearly benign fibroids and malignant leiomyosarcoma exists an intermediate category known as STUMP (Smooth Muscle Tumor of Uncertain Malignant Potential).
These tumors do not meet clear histological criteria for malignancy, yet they cannot be confidently classified as benign. Most STUMPs behave in a non-aggressive manner, although long-term follow-up is recommended.
The existence of STUMP underscores that uterine smooth muscle tumors represent a biological spectrum, rather than a strictly binary classification.
Importantly, the diagnosis of STUMP is usually established only after surgical removal and pathological examination of the tumor, which reflects a broader clinical challenge: accurately determining tumor nature before surgery.
Why is Preoperative Diagnosis So Challenging?
One of the main clinical difficulties is the preoperative differentiation between benign fibroids, STUMP tumors, and leiomyosarcoma. All three tumor types originate in the myometrium, can produce similar clinical symptoms, and may appear comparable on ultrasound or MRI.
At present, no universally accepted preoperative diagnostic method can reliably distinguish benign from malignant myometrial tumors using conventional imaging or routine blood tests alone [2].
This diagnostic uncertainty has important clinical implications, particularly in the context of minimally invasive surgery and power morcellation, where undetected malignancy could affect patient outcomes. Consequently, improving preoperative risk stratification remains an important unmet clinical need.
The Role of Molecular Research
The need for reliable preoperative differentiation between uterine leiomyoma and leiomyosarcoma remains a major clinical challenge.
Recent genomic and transcriptomic studies have identified distinct molecular signatures that differentiate benign from malignant uterine smooth muscle tumors [2]. These differences include mutational burden, copy number alterations, and gene expression patterns.
Molecular research has also clarified key mechanisms underlying fibroid development, including genetic drivers, hormonal regulation, and epigenetic alterations [1].
At the Molecular Diagnostics Group for Gynecologic Tumors at the Carlos Simon Foundation and INCLIVA, research led by Dr. Aymara Mas focuses on translating these findings into non-invasive diagnostic tools. Current work within the team focuses on characterizing tumor heterogeneity and developing liquid biopsy approaches based on circulating cell-free RNA, with the aim of enabling earlier and more reliable preoperative assessment of women with myometrial tumors.
This work is complemented by high-resolution single-cell analyses of the human myometrium, which are helping to refine our understanding of tissue architecture and cellular interactions within the uterus [3].
Together, these efforts seek to improve diagnostic accuracy and support safer surgical decision-making through molecular stratification of myometrial tumors.
References:
- Machado-Lopez A, Simón C, Mas A. Molecular and Cellular Insights into the Development of Uterine Fibroids. Int J Mol Sci. 2021;22(16):8483. Published 2021 Aug 6. doi: 10.3390/ijms22168483
- Machado-Lopez A, Alonso R, Lago V, et al. Integrative Genomic and Transcriptomic Profiling Reveals a Differential Molecular Signature in Uterine Leiomyoma versus Leiomyosarcoma. Int J Mol Sci. 2022;23(4):2190. Published 2022 Feb 16. doi: 10.3390/ijms23042190
- Punzon-Jimenez P, Machado-Lopez A, Perez-Moraga R, et al. Effect of aging on the human myometrium at single-cell resolution. Nat Commun. 2024;15(1):945. Published 2024 Jan 31. doi: 10.1038/s41467-024-45143-z
